Electronegativity periodic table
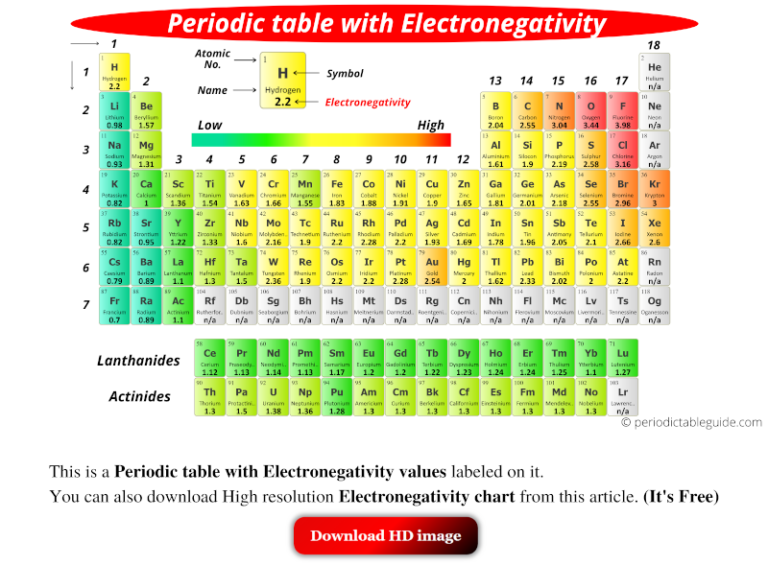

Lord in Macmillan's Chemical and Physical Data, Macmillan, London, UK, 1992.This Electronegativity (Pauling) table gives the Electronegativity (Pauling) of all the elements of periodic table in Pauling Scale. Keiter in Inorganic Chemistry : Principles of Structure and Reactivity, 4th edition, HarperCollins, New York, USA, 1993. These can be found in a variety of chemical textbooks and technical articles as well as online. An electronegativity table of the elements has the elements arranged exactly like in a periodic table, except that each atom is labeled with its electronegativity. in which E (AB) is expressed in kJ mol -1 (1 electron volt, 1eV, 96.48 kJ mol -1) and A - B represents the difference in 'electronegativity' between the two elements, whose individual electronegativities are given the symbols A and. Use an electronegativity table as a reference. The base value of hydrogen was later increased by 0.10 and caesium's electronegativity was later refined to 0.79 however, no refinements have been. Most published medium formulations of the periodic table have a group 3 consisting of: Sc, Y, La and Ac, a sequence that is both logically and chemically incorrect (Thyssen 2011 ). He managed to treat this ionic contribution by the equation. The most electronegative elements are those in the upper right of the periodic table, with the exception of the noble gases. See also: Electronegativities of the elements (data page) The electronegativity of francium was chosen by Pauling as 0.7, close to that of caesium (also assessed 0.7 at that point). 2 generates a periodic table with the four group 3 elements as: Sc, Y, Lu and Ac. Electronegativity (Mulliken-Jaffe -sp 3) The periodic table transformation sequence from Left-Step to the medium form outlined in Fig.Electronegativity (Mulliken-Jaffe -sp 2).Electronegativity (Mulliken-Jaffe - sp).They are non-metals that consist of diatomic molecules. Fluorine and chlorine are the poster children of the halogens. With reference to the alkali metals in Group 1 IA and the halogens in 17 VIIA explain the trend in ionisation potential, electron affinity and electronegativity on moving down the groups in the periodic table. Electronegativity (Mulliken-Jaffe) p-orbital The halogens, aka halogen family, are a group of reactive elements in group 17 of the periodic table, to the right of the chalcogens, and to the left of the noble gases. Thus electronegativity, in general, decreases from top to bottom on moving down a group in the periodic table.You can look at visual representations of the various electronegativity scales using the following links. Tables of values are also given in references 4 and 5 as well. Mulliken's proposals are to be found in references 2 and 3. Values presented here are largely from reference 1. The periodic table’s arrangement also allows scientists to discern trends in element properties, including electronegativity, ionization energy, and atomic radius. Image showing periodicity of the chemical elements for electronegativity (Mulliken-Jaffe) in a 3D periodic table column style. Image showing periodicity of the chemical elements for electronegativity (Mulliken-Jaffe) in a 3D spiral periodic table column style.
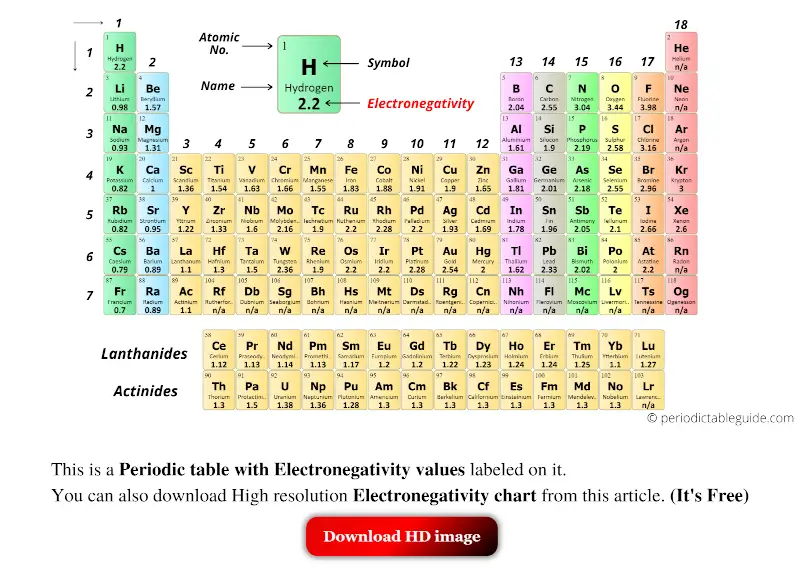
Image showing periodicity of the chemical elements for electronegativity (Mulliken-Jaffe) in a spiral periodic table heatscape style. In the bottom left corner, elements Fr (francium) and Cs (cesium) have values of 0.7. Can you find the lowest value There is no zero shown on the electronegativity chart. Electronegativity is related with ionization energy and electron affinity. Unit for Advanced Study, University of Pretoria, South Africa. The most electronegative atom, fluorine, is assigned a value of 4.0, and values range down to cesium and francium which are the least electronegative at 0.7. Image showing periodicity of the chemical elements for electronegativity (Mulliken-Jaffe) in a periodic table heatscape style. Look at F, fluorine, in the upper right of the periodic table. The higher the associated electronegativity number, the more an element or compound attracts electrons towards it. Electronegativity is a chemical property that describes an atoms ability of attraction on bonded electrons. Χ M = 3.48 Image showing periodicity of the chemical elements for electronegativity (Mulliken-Jaffe) in a periodic table cityscape style. Trend of Electronegativity in periodic table. If the values of IE and EA are in units of MJ mol -1, then the Mulliken electronegativity Χ M can be expressed on the Pauling scale by the relationship: The subscript v denotes a specific valence state - so for trigonal boron compounds, a values of electronegativity can be defined for sp 2 hybrid orbitals. Mulliken proposed an electronegativity scale in which the Mulliken electronegativity, Χ M is related to the electron affinity EA v (a measure of the tendency of an atom to form a negative species) and the ionization potential IE v (a measure of the tendency of an atom to form a positive species) by the equation:
